Imagine yourself in this scenario: in a village, each person is given a stone to build a pathway for their community. The first stone was set by your ancestors, and their only instructions are to place the next stone, including yours, adjacent to another stone. Each stone has its own shape and size. Some are small, and some are large, and some have odd shapes. You might notice that a particular stone seems like it fits perfectly between a stone laid years ago and one that was laid by a friend only months ago; these gaps in the path are waiting for someone to add their stone. The path you see today has twists and turns and branches leading in different directions. When you ask older members of the village, they tell you about the predictions they made when they were young about where the path was headed. You have your own predictions about which direction the path will go. However, each person is free to make their contribution where they think it fits best, building on the work that was done by those before them.
With this village in mind, I want to adjust some details to bring it into the real world. The village represents a community of researchers, clinicians, families, and patients. The first stone was set decades ago by happenstance but is now a landmark for the entire community. Each stone, including my own, is a contribution made over a lifetime. We have our predictions and hopes about the way the path will go, and we are often surprised to find where the path takes us. This village, this community that I have found myself in, is one that involves understanding a rare disease.
Rare diseases (also called Orphan diseases) affect fewer than 200,000 people in the US as defined by the Orphan Drug Act. So for any given rare disease, less than 1 in a thousand citizens are affected. However, there are more than 7,000 rare diseases, and some have only just begun to be described and catalogued, so when combined these rare diseases rival well known diseases in terms of number of people impacted. With proposed budget cuts among other drastic changes to funding of scientific research threatening the work being done, I wanted to take the opportunity to advocate for science funding by telling my story and the story of this community. While the number of people we directly impact might seem small, the work we do is one way we can show our fellow Americans that we care, that we hear their stories, and that we want to help.
My journey started in the fall of 2019, but the beginning of this story dates back decades. Serendipitously, Dr. Gerald Hart was the first to discover a novel modification within the cell, which went against the current understanding within the field. This modification is O-GlcNAc, a sugar added onto proteins within the cell with numerous different roles. This sugar modification is quite unique within the field because of its location inside of cells and the fact that it does not get extended, meaning Dr. Hart was going against all current understanding at the time. We’ve developed our own code to represent these modifications, with O-GlcNAc represented by a blue square. Over the years, a number of the current UGA faculty added to this story including Dr. Robert Haltiwanger and my own advisor Dr. Lance Wells. Dr. Haltiwanger was the first to purify and characterize the enzyme responsible for adding O-GlcNAc onto proteins, O-GlcNAc Transferase (OGT), and Dr. Wells played a central role in characterizing the enzyme responsible for removing the modification, O-GlcNAcase (OGA) both under Dr. Hart’s training. This process is summarized in the picture below. Without decades of public funding, these key first stones may have never been set. Now that we’ve laid the first stones on this path, let us jump forward nearly 2 decades to see where the path is heading.
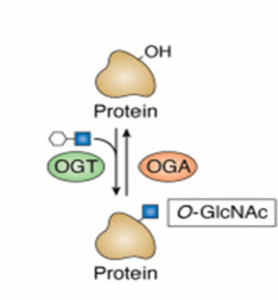
O-GlcNAc is cycled on and off by two enzymes, and it occurs on thousands of proteins. OGT is responsible for adding O-GlcNAc, and OGA is responsible for removing O-GlcNAc from proteins. A simple process with a lot of impact.
Creative commons CC-BY license © 2024 The Authors. Published by Elsevier Inc on behalf of American Society for Biochemistry and Molecular Biology.
In early 2013, Dr. Wells gets a call from the Greenwood Genetic center. Dr. Charles Schwartz is calling because he has identified a patient with intellectual disability and a mutation in the OGT gene. Dr. Wells and Dr. Schwartz start a collaborative effort aiming to understand the role this protein plays in the cell and to understand the rare disease presenting in patients. This effort results in the first characterization of a mutation in OGT that causes intellectual disability. In Europe, another group led by Dr. Daan van Aalten characterize another two families with OGT in 2017. Between 2013 and when I joined the lab in 2019, the collaboration between clinicians like Dr. Schwartz and research labs run by Dr. Wells and Dr. van Aalten would continue to build the path to understanding what is currently called O-GlcNAc Transferase Congenital Disorder of Glycosylation (OGT-CDG). Dr. Schwartz would retire in 2019 just before my part of the story begins, but his career would result in the identification of nearly a third of all genes responsible for intellectual disability on the X-chromosome among other contributions.
Starting in the fall of 2019, I have been working on placing my own stone on the path to understanding this rare disease. I have been working on characterizing novel mutations in the OGT gene to understand the basics of what is wrong at the level of the protein, and I have also been tracking the dozens of mutations that have been reported to us by clinicians across the world. We have been contacted by clinicians frequently from across the globe who are all looking for answers for their patients. You can read about the ins and outs of what we are doing and plan to do, but I want to jump forward to 2024 and talk about the future for this path and highlight one family looking for answers.
With help from an inspirational undergraduate student and the Skripkin family who are looking for answers for their daughter Vica, our laboratory played a central role in helping to organize the first annual conference to understand OGT-CDG in 2024. Cure OGT was set up to bring researchers, clinicians, and families together to discuss the amazing work taking place across the globe, what comes next in order to understand this rare disease, and how the work may translate to the clinic to aid patients and families. The first annual conference had around 45 attendees. Among those in attendance were myself, other graduate students, our own Co-Editor-in-Chief David Steen, post-doctoral fellows, multiple clinicians currently treating patients with OGT-CDG, multiple professors (many of whom were trained by Dr. Hart or Dr. Hanover another founder of the field), the director of the Office of Orphan Product Development via zoom, and most importantly families affected by this rare disease. Over the course of the conference, we discussed what we already know, what we don’t know, and how we move forward. We even heard stories from the families. This community has grown since the first stone on the path was laid, but as you can see the contributions to the future are being laid as we speak by dedicated and caring individuals.
Over 60 families have been identified that have a mutation in OGT leading to intellectual disability in at least one family member since the first in 2013, and while this represents a small population, their lives and their stories matter. Currently, we have neither diagnostics beyond genetic testing and characterization of the mutations, nor do we have approved treatments to help alleviate symptoms. These families deserve answers, and they deserve to have our best and brightest working on this issue. This May, the Skripkin family came to visit the lab to learn more about the work we do here at the Complex Carbohydrate Research Center. Andrey and Irina want to understand OGT-CDG to better understand how it affects their daughter Vica and what options for treatment might be available in the future. Their other daughter, Liza, wants to be a good big sister and that means understanding her sister the best she can. Here is what their family had to offer about why we should fund science and in particular studies investigating rare diseases:
“As a family navigating the daily realities of a rare genetic disease, we know firsthand how isolating and overwhelming it can feel. There are no treatments, little information, and limited help-unless basic research steps in. Investing in fundamental science isn’t abstract for families like ours; it’s the first step towards answers, understanding, and eventually, a cure. We’ve seen inspiring examples where parent-led organizations have helped bring treatments to patients in just a few short years. But what’s easy to overlook is that those “overnight successes’ were only possible because decades of foundational research had already been done. Without that scientific groundwork, no amount of urgency or funding could have moved things forward so quickly. Every discovery made in the lab today lays the foundation for breakthroughs tomorrow-and for families like ours, that means real hope.” -Irina Skripkin
Despite all the progress that has been made, the current administration is jeopardizing current and future science funding. The administration is planning cuts to science funding; it’s making cuts to current research grants and cuts to indirect costs; it has already done away with the fellowship that helped fund my training as a first-generation student and my work on this project leaving future trainees without this type of funding. Building the path forward for families like the Skripkins requires funding, which has historically been a beneficial partnership between universities and the government. In fact, these research dollars have huge financial impacts on the local and national economy and the biomedical industry that has grown up around academic research. In Fiscal Year 2024 (FY24), the National Institutes of Health (NIH) research funding generated $2.56 in economic activity for every dollar spent, according to United for Medical Research (UMR). This translates to $94.58 billion in new economic activity nationwide, supported by nearly half a million jobs.
The work we do not only impacts families like the Skripkins, but it also affects other diseases you might be familiar with. For example, both Eli Lilly and Asceneuron are developing drugs targeting OGA in the hopes of treating tauopathies like Alzheimer’s disease, which affects more than 6 million Americans and is the 7th leading cause of death in the US. These drugs are the direct result of the contributions of scientists and the funding we secure to do the research into this area of inquiry.
You might be wondering how you can contribute to building the path forward for OGT-CDG patients and advocate for science funding. Our own Olivia Asher has written a great article on how to get involved, and she includes her own story of advocating for science funding at the state capitol. You can share this story with others including your representatives, and let them know that the work we do and the work we have done would not be possible without the collaboration between researchers and funding agencies. Or if you or someone you know is affected by a rare disease, you can share your own story to spread awareness.
While I have tried to mention the people who have made this story possible, there are many other undergraduate students, graduate students, post-doctoral fellows, professors, clinicians, and families not directly mentioned who have also each played their part in building the path forward. The work we do and plan to do would not be possible without each of their contributions over the years. I do not know what direction the path will take in the future, but I do know that our contributions today will lead us down a path towards therapeutics and diagnostics for patients like Vica. I want to leave you with a quote from 2022 Nobel prize winner Carolynn Bertozzi and fellow glycobiologist on the purpose of a scientist:
“Your purpose as a scientist is not to achieve fame or money, that is not your purpose… The main goal is to make discoveries and gift them to humanity. And those discoveries and that knowledge stays with humanity long after you’re gone.”
About the Author
Johnathan is a sixth-year Biochemistry and Molecular Biology Ph.D. student. He has bachelor degrees in biology and entomology from the University of Georgia. He also spent two years teaching high school biology and ecology in Nashville, TN before returning to UGA for graduate school. His research focuses on characterizing novel variants of O-GlcNAc transferase that are causal for an X-linked Intellectual Disability. Outside of lab, Johnathan is interested in new TV shows, playing video games, and spending time with his cats, Mowgli and Meelo.





