What comes to mind when you think about the word “physics?â€
You probably see yourself sitting in your high school or college Intro Physics course trying to memorize what seemed to be pointless equations and relationships between variables. For example, the good ole' F=ma. Many of us know these basic physical principles referred to as Newtonian mechanics, after Sir Isaac Newton, the pioneer of the physics of motion. A key characteristic of Newtonian mechanics is that it is deterministic and predictable, meaning we can predict with certainty what will happen next to whatever system we are looking at.
For example, if I tell you certain properties of a box sliding across the floor, you could use Newtonian mechanics to figure out where the box will be in 4 seconds, where it will stop on the floor, etc. But what happens when we try to measure very tiny things, like electrons? If I tell you certain properties of an electron, you would only be able to tell me the probability that it will be at a certain location in 4 seconds.
When we observe very small things, the physics starts to get really weird (Heisenberg's Uncertainty Principle and Schrödinger's Cat come to mind here). At this point, we cannot analyze the electron using Newtonian mechanics; it will not suffice if we want to better understand what is going on. Now, this is where quantum mechanics comes into play!

Unlike Newtonian mechanics, quantum mechanics is probabilistic. This means that when we observe phenomena in the universe on a very tiny scale (on the order of atoms and fundamental particles like electrons), then we can predict the probability that a particle would exist in a certain state, rather than predicting a precise state it will be in when we observe it.
For a concrete example, let's talk about the famous double-slit experiment. When electrons are shot through a wall with two parallel slits onto a screen behind the wall, two things can happen. Sometimes we observe a wave interference pattern on the screen, which suggests that electrons can behave like waves colliding with each other creating ripples. Does this mean electron is really a wave, and not the particle we have been told it is? The short answer is: not really.
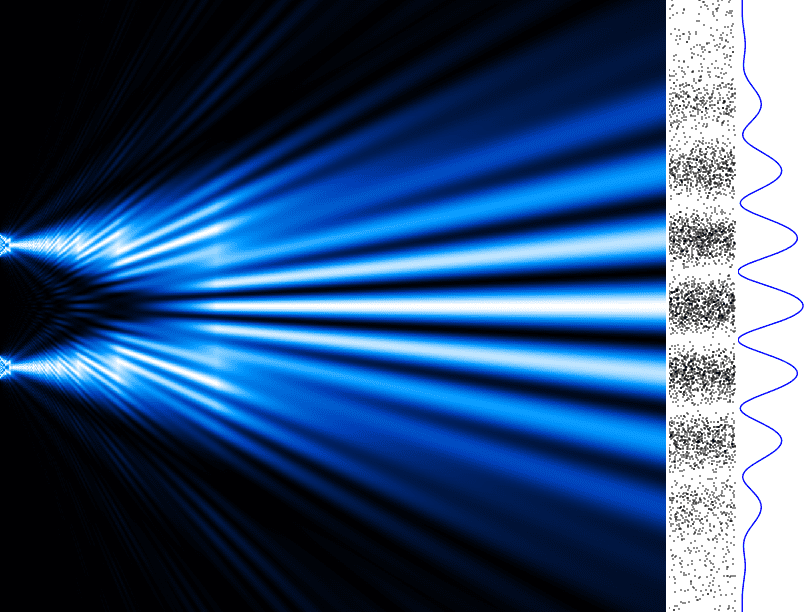
This is where things get really strange. When there are devices placed at the slits that label the electrons based on which slit they entered, the electrons do not produce a wave interference pattern on the screen. Instead, they create a pattern of two parallel lines on the screen where they hit it. But… if we again add another device between the slits and the screen that “erases†the labels that the first device gave the electrons, they will once again produce an interference pattern on the screen like they did when they were not being measured. It may seem like the electrons become aware of being observed and then “decide†to act as particles. This spooky phenomenon is known as the observer effect.
Obviously, the predictions of quantum mechanics are drastically different than the physics we experience in our everyday lives, which are governed by Newtonian mechanics. As objects get larger and larger, the effects of quantum mechanics start to disappear. This is why you would never see a chair existing in two states at the same time, but a small object like an electron is capable of doing this.
Richard Feynman, one of the most influential quantum physicists, sums up the uncertainty of quantum mechanics beautifully in his Messenger Lectures, “I am going to tell you what nature behaves like. … Do not keep saying to yourself, if you can possibly avoid it, ‘But how can it be like that?' because you will get down the drain, into a blind alley from which nobody has escaped. Nobody knows how it can be like that.â€
So the next time you find yourself pondering the nature of the universe and your mind gets boggled by quantum physics, don't be too hard on yourself. Even the best physicists are quite literally incapable of wrapping their minds around the weirdness of the quantum world. For more information about quantum mechanics, stay tuned for part two of my quantum mechanics series!
Cover Image Credit: Apinoid via Flikr Commons
 Paige Copenhaver is an undergraduate studying Physics and Astronomy at the University of Georgia. When she is not building satellites, she can be found playing ukulele or reading Lord of the Rings. Paige also serves on the programming board of Athens Science Café. You can email her at pac25136@uga.edu or follow her on twitter: @p_copenhaver. More from Paige Copenhaver. Paige Copenhaver is an undergraduate studying Physics and Astronomy at the University of Georgia. When she is not building satellites, she can be found playing ukulele or reading Lord of the Rings. Paige also serves on the programming board of Athens Science Café. You can email her at pac25136@uga.edu or follow her on twitter: @p_copenhaver. More from Paige Copenhaver. |







