“Superbugs Strike Again!â€
“Resistance on the Rise!â€
“Antibiotic Resistant Bugs: The Next Pandemic?â€
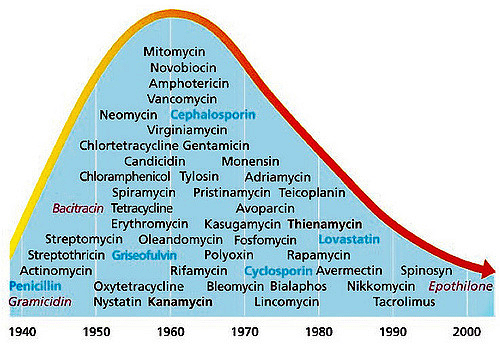
Headlines such as these have become increasingly familiar in the developed world as antibiotic-resistant bacteria, or “superbugsâ€, become more common. Antibiotic resistance occurs when bacteria undergo changes that make them less susceptible to antibiotics; these bacteria cause infections that are difficult to control due to a lack of treatment options. These types of infections, which affect about two million people per year, kill approximately 23,000 people in the United States annually. Antibiotic resistance has been described by The World Health Organization as one of the greatest threats to modern society. As the “golden age†of antibiotics appears to be ending, it is more critical than ever for scientists and physicians to consider treatments other than the “miracle drugs†of the twentieth century.
Antibiotic resistance has led to a kind of “rat race†in the pharmaceutical industry to find the next wonder drug for bacterial infections – a treatment that might change modern medicine like penicillin did in the 1940s. However, despite numerous chemical approaches to solving antibiotic resistance, the answer to this global health crisis may be found outside the laboratory.
Some wild animals are particularly good at fighting off bacterial infections. So, alongside chemical efforts to develop new drugs in the early 2000s, laboratories all over the world began screening wildlife species for antimicrobial peptides, or AMPs – small proteins that can be highly efficient at killing bacteria. Different groups of scientists have identified hundreds of potentially therapeutic AMPs in animals from alligators to beetles. However, perhaps the most promising possessor of potent AMPs are the eight-legged creatures most of us would rather squash than entertain in our homes: spiders.
Spiders are invertebrates that have immune systems far less complex than humans. Instead of having blood contained in vessels, they have open circulatory systems, in which “blood†(called hemolymph), flows freely. Spiders have far fewer varieties of immune cells than humans. However, since spiders are still susceptible to bacterial infections, they mostly defend themselves with small proteins like AMPs. These AMPs can also be found in spiders' silk and venom.

In 2000, Silva and colleagues discovered an AMP from the Brazilian spider Acanthoscurria gomesiana. The researchers termed the protein gomesin after the creature it was derived from. Immediately after its discovery, Silva and colleagues tested the effectiveness of gomesin on a variety of bacterial strains. They found the AMP to be remarkably effective at destroying bacteria. Gomesin was even able to kill fungi and certain parasites, such as Leishmania amazonensis, a causative agent of leishmaniasis, a debilitating infectious disease. Despite this early finding, the antibiotic resistance threat was not as dire in the early 2000s and many forgot about gomesin.
However, in 2017, Henriques and colleagues came across prior studies involving gomesin and noticed something important. The naturally occurring structure of gomesin, shaped much like a hairpin, contributed significantly to its ability to kill bacteria. Therefore, if they could chemically alter gomesin to be even more structurally effective, it could become even more dangerous to bacteria. Henriques and colleagues created a version of gomesin that had a cyclic shape rather than that of a hairpin. They tested this new form of gomesin against various types of bacteria and found it was ten times more effective at killing bacteria than the original form. Since the original form of gomesin was already fairly potent, this was a staggering discovery.
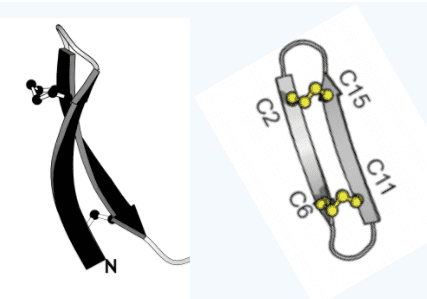
What are the next steps for gomesin and its more effective structural version? The first step is to test these substances' ability to destroy particularly harmful versions of resistant bacteria. For example, researchers are looking at the effectiveness of gomesin and related substances on biofilms, durable, sticky clumps of bacterial cells and materials that can make drug delivery nearly impossible. Additionally, it is important to understand exactly how these kinds of substances destroy bacteria to make sure they will not also harm patients in the process. Finally, if gomesin achieves success in trials using animal models, the substance could be tested for use in humans. Because of spiders, we may have unlocked an important weapon for an alternative arsenal against resistant bacteria.
Perhaps next time you are frightened by one of our eight-legged friends, reconsider going for the squash. It is possible we will all be resorting to spider-derived AMPs such as gomesin to treat nasty cases of strep throat in the future.
About the Author
 Jenn is an MS student in the Department of Infectious Diseases at the University of Georgia, where she studies immune trade-offs in tissue-regenerating rodent species. Jenn loves to dance, draw, hike, and demolish large quantities of food in her spare time. She's also in love with dogs and spends plenty of time with her pampered pooch, Ziggy. Feel free to check out a few of her ASC poster designs, personal research website, or to contact her at jennifer.cyr25@uga.edu or on Twitter at @jennlcyr! Jenn is an MS student in the Department of Infectious Diseases at the University of Georgia, where she studies immune trade-offs in tissue-regenerating rodent species. Jenn loves to dance, draw, hike, and demolish large quantities of food in her spare time. She's also in love with dogs and spends plenty of time with her pampered pooch, Ziggy. Feel free to check out a few of her ASC poster designs, personal research website, or to contact her at jennifer.cyr25@uga.edu or on Twitter at @jennlcyr! |







