Over the course of the year 2005, nearly 9,500 people lay in bed with arsenic coursing through their veins. They had watched as the toxic drug was slowly forced into their bloodstream, hopefully through a glass syringe; as it is dissolved in propylene glycol, a close relative of antifreeze, the drug melts plastic syringes used for injections.

Lying in the stifling hospital ward, muscles clenched in protest of the fire in their veins, they began to think that perhaps the convulsions, confusion and lapses in consciousness characteristic of their affliction are not the worst a person can endure. And yet without the drug, sleepless nights and lifeless days would fade into coma and ultimately prove fatal.
A spinal tap days before revealed that their cerebrospinal fluid contained Trypanosoma brucei – the protozoan parasite responsible for causing sleeping sickness, or Human African Trypanosomiasis (HAT). Parasites invaded their bodies weeks ago with the bite of a tsetse fly. By now parasitic microbes called trypanosomes had penetrated the blood brain barrier, traversing unknown paths into the fluid that cushions and nourishes the brain.
After as much as a month of treatment, the drug, Melarsoprol, saved only 7,410 of those treated. The sleeping sickness overcame 1,852, a single mutation in the parasite giving it all it needed to resist treatment. For the remaining 238, though treatment could not save them, it wasn't the parasites that killed them; Melarsoprol causes swelling and inflammation of the brain in 5-10% of patients, and of those, half do not survive.
Rights versus realities
“The enjoyment of the highest attainable standard of health is one of the fundamental rights of every human being without distinction of race, religion, political belief, economic or social condition.”
So says the Constitution of the World Health Organization (WHO), drawn up by the International Health Conference in 1946. So how is it that as recently as 2008, a drug like Melarsoprol – highly toxic and susceptible to the development of drug resistance – was still the first line drug for the treatment of late-stage Human African Trypanosomiasis? Why, in the 60 years since Melarsoprol's introduction, have researchers not been able to develop a drug that doesn't kill one out of every 20 people it's trying to save? Is that really the “highest attainable standard of health?â€
The question is a complicated one that spans not just the treatment and control of Human African Trypanosomiasis, but that of all diseases that disproportionately afflict countries in the developing world. “Compared to a lot of other problems, they're still vastly neglected,†says Dr. Dan Colley of the University of Georgia Center for Tropical and Emerging Global Diseases. “Because many of them are chronic infections that don't kill people, because they don't have the obvious DALYs [disability-adjusted life years] of some diseases, because they're diseases of the poor — in the countries where they're endemic, they're often ignored.â€
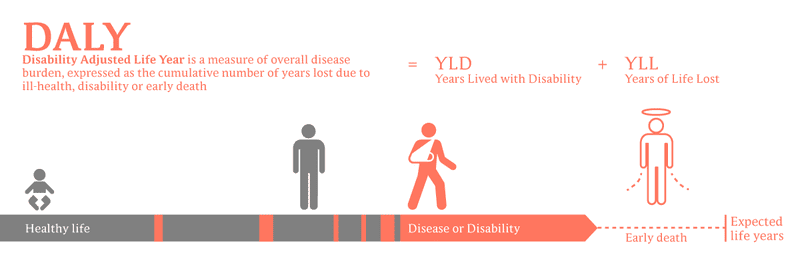
Access to health care is a basic human right; as such, the gross health disparities that exist across the globe, particularly between the developing and developed regions of the world, should be of concern to all countries, not just those who bear the heaviest burden of disease. In an ideal world, humanitarian concerns would dominate conversations surrounding funding decisions. For better or worse, however, we do not live in such a utopian society.
Access to health care is a basic human right; as such, the gross health disparities that exist across the globe should be of concern to all countries
Historically, national interests and profitability have played a significant role in the response to global and public health concerns. Colonial interests largely dictated the United Kingdom's tropical disease research agenda in the 19th century. Though chloroquine was identified as an effective antimalarial in 1937, the United States did not begin clinical trials to bring it into widespread use until the return of soldiers from the Pacific Theater of World War II prompted concerns about malaria in the United States.
Dr. Christopher Whalen, the Ernest Corn Professor of Infectious Disease Epidemiology at the University of Georgia, recalled that in the early days of the AIDS pandemic, “For three or four, almost five years, we had protease inhibitors. They were prohibitively expensive here [in the United States], they were unattainable in Africa.†To help in the fight against HIV, the drug companies were able to donate small amounts of protease inhibitors, which block the activity of viral proteins critical for causing disease, but could not afford to hand out the drug in the large quantities that were desperately needed in Sub-Saharan Africa at that time. The only way to get the drugs to the populations that needed them was to make low- and middle-income countries more attractive markets.
“One of my colleagues brilliantly used the generic and proprietary drugs for HIV, and played them off one another to drive down the cost of proprietary drugs in Africa,†recalls Whalen. “And he made the point – he said to the drug companies, ‘Where's the largest market for your drug? Sub-Saharan Africa.'†The colleague in question was a pediatrician who treated a large number of AIDS patients at that time. His reasoning was that a drug sold at a significantly reduced price still makes more money than a drug sitting untouched in a warehouse until it expires. “He did not accept the global health disparities as a reason for why HIV couldn't be treated in Africa. So he very persistently tried to get the drug.â€
Even the use of Melarsoprol for HAT treatment was driven by concern for costs. In the mid-1980s, an alternative drug to Melarsoprol, eflornithine, was shown to be effective against late-stage HAT. In fact, it was so effective that it was nicknamed “the resurrection drug†for its ability to raise comatose patients from their corpse-like state. Originally developed as an anti-cancer medication, eflornithine was tested in clinical trials and exhibited a good safety profile. However, in the end it failed to effectively fight cancer and its production ceased entirely in 1999.
That same year, the company that had been producing eflornithine, the predecessor to current French pharmaceutical giant Sanofi-Aventis, sold the WHO the license for eflornithine along with all of its remaining stocks. With the help of Médecins Sans Frontières (Doctors Without Borders, MSF), the WHO tried – and failed – to find an alternative manufacturer for eflornithine. Because drugs that treat diseases affecting poor, relatively small populations generate little profit, it was not economically viable for pharmaceutical companies to manufacture eflornithine.
In 2000, eflornithine was given a second chance when the FDA approved its use in Vaniqa, a new topical cream used for, of all things, hair-removal. The issue found its way into the public eye when early in the following year, 60 Minutes aired a heart-rending report by Christiane Amanpour on the horror of late-stage HAT infection in South Sudan. The American public watched as grown men receiving Melarsoprol injections wept, and a 12-year-old boy in the late stages of infection fell unconscious mid-conversation. A young child being treated with Melarsoprol cried out, “Mommy, mommy, why are you hurting me?†For added effect, 60 Minutes ran the advertisement for Vaniqa near the end of the segment.
The moral outrage that ensued was fanned furiously by the MSF in the hopes that bad publicity would induce the pharmaceutical industry to rethink their stance on eflornithine production. No companies were willing to produce the drug to help save thousands in Sub-Saharan Africa from a usually fatal disease, but as a beauty product predicted to earn $200 million USD in sales its first year? Sure, why not. In May 2001, public pressure helped the WHO sign a deal with Aventis to ensure the continued production of eflornithine for the treatment of HAT.
The story didn't end there, though; Melarsoprol remained the first-line drug for treatment of HAT for the better part of a decade, even after eflornithine was available. Safe use of eflornithine requires a long course of treatment and additional clinical care; a patient undergoing eflornithine treatment requires a minimum 10 day hospital stay with supportive care, though treatment can last over a month. This requires funding and trained medical personnel, rendering eflornithine treatment too costly and logistically complex to provide on a large scale to communities that have little or no money to pay for it.
“Big Pharmaâ€
Lest righteous indignation take over and we indiscriminately vilify pharmaceutical companies, let’s consider the practical issues surrounding neglected disease control.
While pharmaceutical companies are in a prime position to do good by providing much-needed medications, they are not philanthropies; they are businesses with shareholders to keep happy and profit margins to consider. If pharmaceutical companies fail in their business obligations by investing large sums of money in ventures that will return no profit, they risk collapsing or, more realistically, being out-competed by other companies.
While pharmaceutical companies are in a prime position to do good by providing much-needed medications, they are not philanthropies
Interfering with a business-centric model in favor of a more philanthropic one is economically unviable, and may have unforeseen consequences. “I think at that point you probably lose competition, lose the thinking that goes along with that competition, and maybe not have the drugs that you need to develop,†says Whalen. “It is hard when you have a humanitarian emergency, however, to know that there's a company that is unwilling to provide its drug for whatever reason,†he added.
Pharmaceutical companies are the third largest funder of research and development for neglected tropical diseases. Many aid organizations, including the United States Agency for International Development (USAID) and the WHO, rely on donations of drugs from these companies for control efforts in low-income communities. USAID estimates that the pharmaceutical industry has donated over $3.1 billion USD in drugs to fight neglected tropical diseases. If companies are unwise in their business practices, they will be less able to provide much-needed money and resources to ongoing aid programs.
On top of that, the cost of developing a drug can be astronomical. While the figure is highly contested, a 2014 study out of the Tufts Center for the Study of Drug Development estimates that the cost of developing a single drug from screening to approval for clinical use is approximately $2.56 billion USD. This cost includes not only the expenses directly involved in taking a drug from compound screening, through preclinical laboratory tests and clinical trials, and finally to approval by regulatory agencies; it also includes the money spent on the multitude of failures that are an essential part of all scientific discovery.
Do these costs and practical considerations justify the use of a treatment, like Melarsoprol, that is dangerous and of questionable efficacy? That is a subject for a different forum; however, they reinforce the notion that the issues at hand are not black-and-white or right-versus-wrong. In order to fulfill our moral obligation to our fellow man while living within the practical constraints of the real world, we need to approach neglected disease control in a thoughtful and creative manner.
Optimizing efforts
The development of new scientific tools for use in medicine and other industries requires several levels of research. The first is basic research, through which scientists seek to expand the breadth of current scientific knowledge without a specific treatment or clinical application in mind.
Promising therapeutic or diagnostic targets identified during basic research are brought forward to “translational research,†often referred to as a “bench-to-bedside†approach. Translational research advances the discoveries made during basic research from the realm of the academically interesting to that of the clinically relevant by developing them into new tools, diagnostic techniques, and therapies that can be used in a practical setting.
Novel medical tools developed during translational research then need to undergo clinical development, which entails extensive testing in a clinical setting to evaluate a tool's safety and efficacy. Clinical development is essential for regulatory agencies to obtain sufficient information to justify approving a drug or technique.
Once a therapy or diagnostic technique is developed, it is essential to determine the most effective way to implement it. Though clinical development has determined the safest way to administer this new tool to a population, has it also determined the best way?
In 1975, the WHO formed the Special Program for Research and Training in Tropical Diseases (TDR.) This program was designed with the understanding that no public health problem can be solved simply by throwing drugs and money at it, even if sufficient resources existed to do so. In order to control a disease in an effective and meaningful way, aid organizations need to understand the social, economic, and environmental factors surrounding a disease.
TDR's application of social and economic study is one of many examples of operational research and implementation research, the goals of which are to maximize the effectiveness and sustainability of global public health programs while minimizing associated costs and risks. These studies assess many varied facets of tropical disease control efforts, from how treatments can be administered to the greatest effect, to understanding how to engage a community in control and prevention efforts. Additionally, protocols need to be adapted to fit the social context in which they will be implemented.
Operational and implementation research programs help to streamline disease control efforts. In 2001, the WHO established guidelines for the treatment of schistosomiasis, a worm infection that causes chronic disease in at least 240 million people in tropical regions worldwide. These guidelines state that in order to control schistosomiasis morbidity, drugs should be administered primarily to primary school-age children annually or every other year. The children would likely get the disease again, but following such a treatment program should prevent them from being seriously impacted by it. While the literature and extensive reworking by experts guided the design of these guidelines, in reality, there was not a lot of empirical evidence to support them.
Thus, in 2008, the Schistosomiasis Consortium for Operational Research and Evaluation (SCORE) was established with Dr. Dan Colley at the helm to perform operational research on schistosomiasis control. With funding from the Bill and Melinda Gates Foundation, SCORE asks questions on the level of how to control schistosomiasis morbidity, how to sustain that control, and then how to eliminate the disease.
SCORE approaches these questions by assessing the effectiveness of current schistosomiasis diagnostic tools and treatment guidelines in a clinical setting. “We have field-based operational research questions and we have diagnostic questions, because the diagnostic tools used for schistosomiasis aren't very good,†explains Colley. “They've served us well for 40 years but they are not terribly sensitive, and when you get down to treating people every year or every other year, you need sensitive tests because you have knocked [schistosomiasis incidence] down a long way.â€
“The drug supply currently isn't the biggest problem. The biggest problem is how do you use it most effectively?”
SCORE also runs large field studies in five African countries to test drug delivery schemes. The drug for schistosomiasis, praziquantel, is provided by the manufacturer to the WHO, who distributes it to national control programs. “So the drug supply currently isn't the biggest problem,†says Colley. “The biggest problem is how do you use it most effectively? Should the drug be delivered every year, should it be delivered through schools, should it be delivered through communities, should it be delivered through both? Can you skip a year? Some of the guidelines say you can do every other year. Does that work as well or almost as well as doing it every year?†If SCORE finds that drugs can be delivered less often to smaller groups, it means that the same resources can go much further.
Schistosomiasis is transmitted when a person bathes in fresh water containing schistosome-infected snails. Schistosomes infect these snails in the first place when feces or urine, depending on the species of schistosome, containing the parasite is dumped into the water. Thus, keeping people out of infected water is critical, but how do you do that?
SCORE addresses this issue through “sensitization†of the community to the schistosomiasis problem – helping the community understand that the disease is a problem, teaching them what the SCORE program hopes to accomplish, and getting them involved in the work. “The challenges in schistosomiasis control or elimination would come in a couple of different ways, and certainly the behavioral change is a major part of the work,†says Colley, particularly referencing SCORE's work on the island of Zanzibar, off the coast of Tanzania.
Social scientists working with SCORE hold “Kichocho Day†– “Kichocho†is the Swahili word for urogenital schistosomiasis – in local primary schools to educate children on schistosomiasis and how it is spread. “The schools invent games, they have story times, they do all sorts of things,†says Colley. “They're very much involved.â€
Teaching people how to avoid disease and not giving them an alternative is pretty fruitless.
Teaching people how to avoid disease is not enough, however. “Telling people and explaining to people and not giving them an alternative is pretty fruitless. ‘Don't go in the water! Don't go in the water! Don't go in the water!' But there's no other water. That doesn't work.†A problem encountered in schistosomiasis studies and control efforts around the world was that of women becoming infected while washing clothes in schistosome-infected water sources. In response, SCORE has built washing platforms so the women can wash clothes in a social setting, while avoiding exposing themselves to the parasite.
“But what would the women on Zanzibar think a good washing platform looks like?†asks Colley. “Well, you involve them in focus groups and they tell you. ‘No, no, no, we want something that we can all sit around in a circle.' Ok, so let's think about this design. Well, what design is cheap enough that we can do it with local materials? All of those sorts of things have to be done to get something effective.â€

Consideration of social needs in addition to health concerns and providing a reasonable alternative to unhealthful behavior are crucial to schistosome control efforts. “You do those things you have to do to get people to take up what you think they ought to be doing. And sometimes you change somewhat what you think they ought to be doing.â€
Too much of a good thing?
Some basic science researchers are concerned that too much emphasis is being placed upon operational and implementation research. “I think you have to be careful, because if we did nothing but operational research, we'd still be using an Iron Lung to treat polio,†says Whalen. He is one of many concerned that too many operational and implementation research programs draw funding from basic research projects, at the risk of impeding discovery of new tools and therapeutic targets.
“You need the operational research to introduce and test a new idea at a population level, but you can't just turn off the switch and say, ‘How can I do this particular activity better?' That's good, but if you're just saying, ‘How do I do Iron Lung treatment of polio better?' and you don't develop a vaccine or, for other diseases, you don't develop the antiviral or the antibacterial, you end up perpetuating the disease in a sense.” Whalen emphasized the need for research at all levels from the ground up. “There are those way upstream things that if you cut them off you may miss those discoveries, so it's a balance.â€
Colley is less concerned. What offsets this concern, he says, is that the funding for these different types of research comes from different places. As a result, basic research and operational or implementation research are not in competition; they're complementary. “The NIH is going to fund basic science on these things, and the [Bill and Melinda] Gates Foundation is only going to fund, in the strictest sense, some basic science, but mainly operational research, implementation kinds of things. USAID is going to fund mainly implementation and a little bit of operational research.â€
Colley, however, agrees with Whalen on the need for new tools to effectively tackle tropical infectious diseases. “Hopefully the basic sciences will help us come up with new answers, because we certainly don't have the perfect drugs, we don't have the best diagnostics, we don't have the best vaccines, or really any vaccines, so those still need to be developed,†says Colley. “At the same time, we have tools that should be implemented for the people who are suffering right now. So you need to do the whole spectrum.â€
The beginning of a beautiful friendship
Partnerships between the public, private, and philanthropic sectors have proven essential to most effective control efforts. A 2001 MSF study showed that of all the new drugs developed between 1975 and 1999, only 1.1% (a total of 16 drugs) were specifically approved for the treatment of tropical diseases, despite the fact that these diseases made up approximately 12% of the global health burden.
The result of this study was a global shift in tropical disease research and development with the rise of not-for-profit product development partnerships, or PDPs. The WHO teamed up with academic institutions, pharmaceutical companies, philanthropic organizations and governments from both the developing and developed world to initiate programs such as the Program for Appropriate Technology in Health, the International AIDS Vaccine Initiative, the Medicines for Malaria Venture and the Global Alliance for Tuberculosis Drug Development, among many others.
A significant driving force behind this shift was the Bill and Melinda Gates Foundation, which was established in 2000. Donating $8 billion USD to global health programs, they became the largest non-governmental source of funding for tropical disease control and research. “Without that financial support,†said Colley, “all those other partners would have had a very hard time coming together.â€

While such partnerships are largely advantageous, they are not perfect. Difficulties arise when coordinating so many people and organizations with different agendas, regardless of the overall goals of the partnership in question. “You have to deal with people as well as ideas, and so there are always glitches, there are always changes in programs,†observes Colley, “but by and large, anyone that's been in this business for 45 years like I have sees that we're in a whole lot better place than we ever were. That doesn't mean it's perfect by any means, and that doesn't mean it won't continue to evolve and develop.â€
Since 2000, the efforts of these partnerships have resulted in great global health achievements. Between 2000 and 2009, child mortality, 99% of which occurs in low- to middle-income countries like those in Sub-Saharan Africa and Southeast Asia, dropped from 77 per 1,000 live births to 62 per 1,000.
During this same time frame, mass vaccination programs averted an estimated 12.7 million deaths due to measles, 700,000 annual deaths due to hepatitis B-induced cirrhosis and liver cancer, and 130,000 annual deaths due to pneumonia and meningitis in children under 5 years old. 800 million people obtained access to clean water sources, and 570 million more to improved sanitation.
Yet for all that progress, much work remains to be done. While malaria mortality rates have dropped by an estimated 47% since 2000, 584,000 people still die from malaria each year, primarily children under the age of 5 in Sub-Saharan Africa. Of the 7 billion people living on Earth, 2.5 billion still do not have access to adequate sanitation. These partnerships need to continue to evolve if progress is to continue; stagnant practices and fixed protocols can only serve to impede further advancement.
The cost of a cure
In 2003, with the help of TDR and five public sector institutions, MSF established a not-for-profit Product Development Partnership, the Drugs for Neglected Diseases initiative (DNDi). DNDi has been highly effective at showing that drug development need not be a multi-billion dollar enterprise.
Between 2003 and 2013, DNDi delivered six new drugs for the treatment of neglected tropical diseases with 12 more in development, though expenses totaled only about €182.5 million (approximately $239.7 million USD). Even considering that an additional 20% of would-be expenses are covered by in-kind donations, like the donation of drugs by pharmaceutical companies for use in clinical trials, this number is a far cry from the $2.6 billion USD per drug estimate of the Tufts study.
Despite the progress being made in tropical disease R&D efforts, there is still much room for improvement. A follow-up to MSF’s 2001 study that was performed in 2012 showed that of the 756 new drugs approved for human use between 2000 and 2011, 3.8% were for tropical diseases (the global burden at that time had dropped to 10.5%). Further collaborations to streamline and reduce the costs of drug development will be necessary to increase the number of safe, effective, and widely available treatments for neglected diseases.
Rather than developing new compounds from scratch, drug repurposing tests drugs that have already been proven safe in clinical trials in a new application.
One key aspect to the success of DNDi in keeping development costs low is the idea of drug repurposing. Rather than screening new compounds that have to be developed from scratch, drug repurposing tests drugs that have already been proven safe in clinical trials in a new application. This tremendously reduces cost and time spent developing a drug; all that remains are clinical trials to test the efficacy of the drug and to confirm its safety in a new context. While performing clinical trials still requires a significant investment of time and resources, cutting out the preclinical stages of development enables some repurposed drugs to be approved in as little as 3 years, compared to the 10-15 years it takes to develop a new drug from nothing.
DNDi used drug repurposing to great effect in the fight against late-stage HAT. DNDi tested a combination therapy of eflornithine and nifurtimox, a drug already used in the treatment of HAT's New World cousin, Chagas Disease. As this nifurtimox-eflornithine combination therapy (NECT) only needed to undergo Phase III clinical trials and implementation, it was developed in just under 6 years, making the WHO essential medicines list in 2009.
By 2012, 96% of late stage HAT cases were treated with NECT, replacing the toxic “arsenic-in-antifreeze†drug, Melarsoprol, as the first-line treatment for late-stage HAT. Even this Cinderella story has a catch, however; NECT is only effective against Trypanosoma brucei gambiense, the more common of the two subspecies of parasite that causes HAT.
Drug repurposing also has great potential to provide adjunctive therapies to treatments already in use. Current research seeking to understand the mechanisms underlying the pathogenesis of tropical diseases may help to reveal a host's contribution to disease.
An understanding of disease from a host response perspective allows medical professionals to fight symptoms and indirect effects of infections in addition to killing the pathogen directly. This creates an opportunity to attack a disease from several fronts, and opens up the possibility that inexpensive, widely used medications such as anti-inflammatories and anticoagulants may be effective at preventing morbidity and mortality when used in combination with existing antiparasitic and antibiotic drugs.
A vested interest
Heavy disease burden can cripple a local economy; employee illness decreases productivity, and increases absenteeism and company health care spending. In malaria-endemic countries, reports estimate that an employee will miss between 1-10 days of work for each bout of malaria, and exhibit significant reductions in productivity upon their return.
Some companies in these regions are not waiting for external aid organizations to solve this problem. In one study, three companies in Zambia provided malaria control services for their employees and dependents, particularly indoor residual spraying and the distribution of insecticide-treated nets. The result was a substantial drop in annual malaria cases seen in the company clinic from 27,925 to 1,631, and a corresponding 94% decrease in work days lost due to malaria illness. These malaria control services produced at least a 28% rate of return on the companies' investments, even according to the most conservative estimates.

This study and others like it show that aid organizations, governments and pharmaceutical companies are not the only ones in a position to play an important role in the fight against tropical diseases. If employers in disease-endemic regions provide improved employee health care, they stand not only to improve regional public health, but also to see direct economic gains from their investment.
Looking to the future
The successes of neglected disease research and control programs in the last decade have shown us that while balancing humanitarian and practical concerns is a delicate and complex endeavor, it's not impossible. Partnerships between public and private sectors around the world and research-directed public health programs have made slow but important steps toward control of some tropical diseases, notably HAT and malaria.
Though a balancing of global health disparity is still distant, continued dedication to a proactive and coordinated global health agenda will yield real progress towards closing the gaps in regional health status. With enough time, funding, and luck, this work promises to help the developing world achieve the “highest attainable standard of health†that has always been their fundamental, if seemingly unobtainable, right.
About the Author
 Tara Bracken is a Ph.D. candidate at the University of Georgia Department of Infectious Diseases specializing in Global Health. Tara's research focuses on how the host's response to disease worsens malaria infection in children under the age of five and pregnant women. A true graduate student at heart, Tara is roughly 55% water and 45% caffeine. If you're a nerd and like fun, be sure to check out her blog, Of Microbes and Men, or you can connect with her on Twitter at @microbesandmen. Tara Bracken is a Ph.D. candidate at the University of Georgia Department of Infectious Diseases specializing in Global Health. Tara's research focuses on how the host's response to disease worsens malaria infection in children under the age of five and pregnant women. A true graduate student at heart, Tara is roughly 55% water and 45% caffeine. If you're a nerd and like fun, be sure to check out her blog, Of Microbes and Men, or you can connect with her on Twitter at @microbesandmen. |
References Cited
Balasegaram, M., Harris, S., Checchi, F., Ghorashian, S., Hamel, C., & Karunakara, U. (2006). Melarsoprol versus eflornithine for treating late-stage Gambian trypanosomiasis in the Republic of the Congo. Bulletin of the World Health Organization, 84(10), 783–791.
Barrett, M. P. The rise and fall of sleeping sickness. (1474-547)
Beachy, S. (2014). Introduction and Themes of the Workshop. In Drug repurposing and repositioning: Workshop summary. Washington, D.C.: The National Academies Press.
Drugs for Neglected Diseases Initiative. (Jan 2014). An Innovative Approach to R&D for Neglected Patients: Ten Years of Experience & Lessons Learned by DNDi. http://www.dndi.org/images/stories/pdf_aboutDNDi/DNDiModel/ DNDi_Modelpaper_2013.pdf
Eperon, G., Balasegaram, M., Potet, J., Mowbray, C., Valverde, O., & Chappuis, F. Treatment options for second-stage gambiense human African trypanosomiasis. (1744-8336).
Fèvre, E. M., Wissmann, B. v., Welburn, S. C., & Lutumba, P. (2008). The Burden of Human African Trypanosomiasis. PLoS Negl Trop Dis, 2(12), e333.
International Federation of Pharmaceutical Manufacturers & Associations. (2014). Pharmaceutical R&D Projects to Prevent and Control Neglected Conditions. http://www.ifpma.org/fileadmin/content/Publication/2015/IFPMA_ 2014_Status_Report_NTDs_FINAL.pdf
Pécoul, B., Chirac, P., Trouiller, P., & Pinel, J. (1999). Access to essential drugs in poor countries: A lost battle? JAMA, 281(4), 361-367.
Peters, D. H., Adam, T., Alonge, O., Agyepong, I. A., & Tran, N. (2013). Implementation research: what it is and how to do it. Journal Article BMJ (Vol. 347).
Pharmaceutical Research and Manufacturers of America. (Feb 2007.) Drug Discovery and Development: Understanding the R&D Process.
Roll Back Malaria Partnership. (2011). Business Investing in Malaria Control: Economic Returns and a Healthy Workforce for Africa. (Progress & Impact Series, n. 6). http://www.rollbackmalaria.org/microsites/ProgressImpact Series/docs/report6-en.pdf
Schistosomiasis Consortium for Operational Research and Evaluation (SCORE): http://score.uga.edu
Simarro, P. P., Franco, J., Diarra, A., Postigo, J. A. R., & Jannin, J. (2012). Update on field use of the available drugs for the chemotherapy of human African trypanosomiasis. Parasitology, 139(07), 842-846.
Special Programme for Research and Training in Tropical Diseases. (2007). Making a Difference: 30 Years of Research and Capacity Building in Tropical Diseases. http://www.who.int/tdr/publications/documents/anniversary_ book.pdf?ua=1
Sommerfeld, J., Ramsay, A., Pagnoni, F., Terry, R. F., Guth, J. A., & Reeder, J. C. (2015). Applied Research for Better Disease Prevention and Control. PLoS Negl Trop Dis, 9(1), e3378.
Tkach, A., & Amanpour, C. (2001). Sleeping Sickness [Television series episode]. In 60 Minutes. CBS. URL: https://www.youtube.com/watch?v=LfeMIcWK3oQ
Ten great public health achievements–worldwide, 2001-2010. (2011). CDC Morbidity and Mortality Weekly Report, 60(24), 814-818.
Trouiller, P., Torreele, E., Olliaro, P., White, N., Foster, S., Wirth, D., & Pécoul, B. (2001). Drugs for neglected diseases: a failure of the market and a public health failure? Tropical Medicine & International Health, 6(11), 945-951.
Tufts Center for the Study of Drug Development. (2014, March). Cost of Developing a New Drug. http://csdd.tufts.edu/news/complete_story/cost_study_press_ event_webcast
United States Agency for International Development. (2012, September). Neglected Tropical Diseases. http://www.usaid.gov/news-information/fact-sheets/neglected-tropical-diseases
United Nations International Year of Water Cooperation: Facts and Figures http://www.unwater.org/water-cooperation-2013/water-cooperation/facts-and-figures/en/
Wolburg, H., Mogk, S., Acker, S., Frey, C., Meinert, M., Schönfeld, C., . . . Duszenko, M. (2012). Late Stage Infection in Sleeping Sickness. PLoS ONE, 7(3), e34304.
World Health Organization. (2007). Application For Revision Of Antitrypanosomal Medicines In WHO Model List Of Essential Medicines. http://archives.who.int/eml/expcom/expcom15/applications/ sections/Antitrypanosomal.pdf
World Health Organization. (2014.) 2014 World Malaria Report. http://www.who.int/malaria/publications/world_ malaria_report_2014/wmr-2014-no-profiles.pdf
World Health Organization. (2015.) Contribution of pharmaceutical companies to the control of neglected tropical diseases. http://www.who.int/neglected_diseases/Donation_ table_2012.pdf?ua=1







