Coughing, sneezing, runny nose, yes it's that time of year again—flu season is upon us. By now you've seen plenty of “get your flu shot here†paraphernalia. Each year, a new flu shot is developed to try to combat the virus strains predicted to wreak havoc that season.

As we learned in Part II of the influenza blog installment, the mutation and reassortment capabilities of influenza viruses result in a constantly evolving genetic pool of material in multiple species.
Sound complicated? It is.
Flu vaccines stimulate antibodies to develop in the body about two weeks after you get your shot. These antibodies provide protection against infection from the viruses that are in that particular vaccine. But remember, many different combinations of hemagglutinin (HA) and neuraminidase (NA) proteins on the surface of the flu virus are possible. Recall Part I of the influenza blog installment where we discussed two flu strains linked to canine influenza outbreaks.
So while your body remembers the virus, you still need to get a flu shot every year because the flu strains differ from year to year and immunity declines over time.
So, who decides which viruses make it into the flu vaccine?

All year long, 142 national influenza centers in 113 different countries collect data on the flu viruses making people sick. All of these smaller centers then pass representative viruses identified by this global network of laboratories to one of five World Health Organization (WHO) Collaborating Centers for Reference and Research on Influenza. Scientists at each of the five main centers then gather and analyze the data together to identify new flu strains, how efficiently those strains are spreading, and how well previous vaccine strains have worked to combat their targeted viruses. Twice a year the WHO organizes a meeting with the Directors of the WHO Collaborating Centers, who review the results and make recommendations on the composition of the seasonal influenza vaccine. Every flu shot contains protection against three or four strains.
Another factor that helps determine which strains of the flu will be included in the upcoming seasonal flu shot is the ability to produce a working vaccine against that particular strain. In the U.S., the Food and Drug Administration (FDA) makes the final decision about vaccine viruses included in the upcoming season's flu vaccine. Every vaccine must be thoroughly tested and approved by the FDA before it is made available to the public.
So once they are approved, how do these flu vaccines get manufactured?
While the FDA signs off on the strain composition for the new vaccine, private sector manufacturers make all commercially available flu vaccines in the U.S. There are three different influenza vaccine production technologies approved by the FDA: egg-based, cell-based, and recombinant.
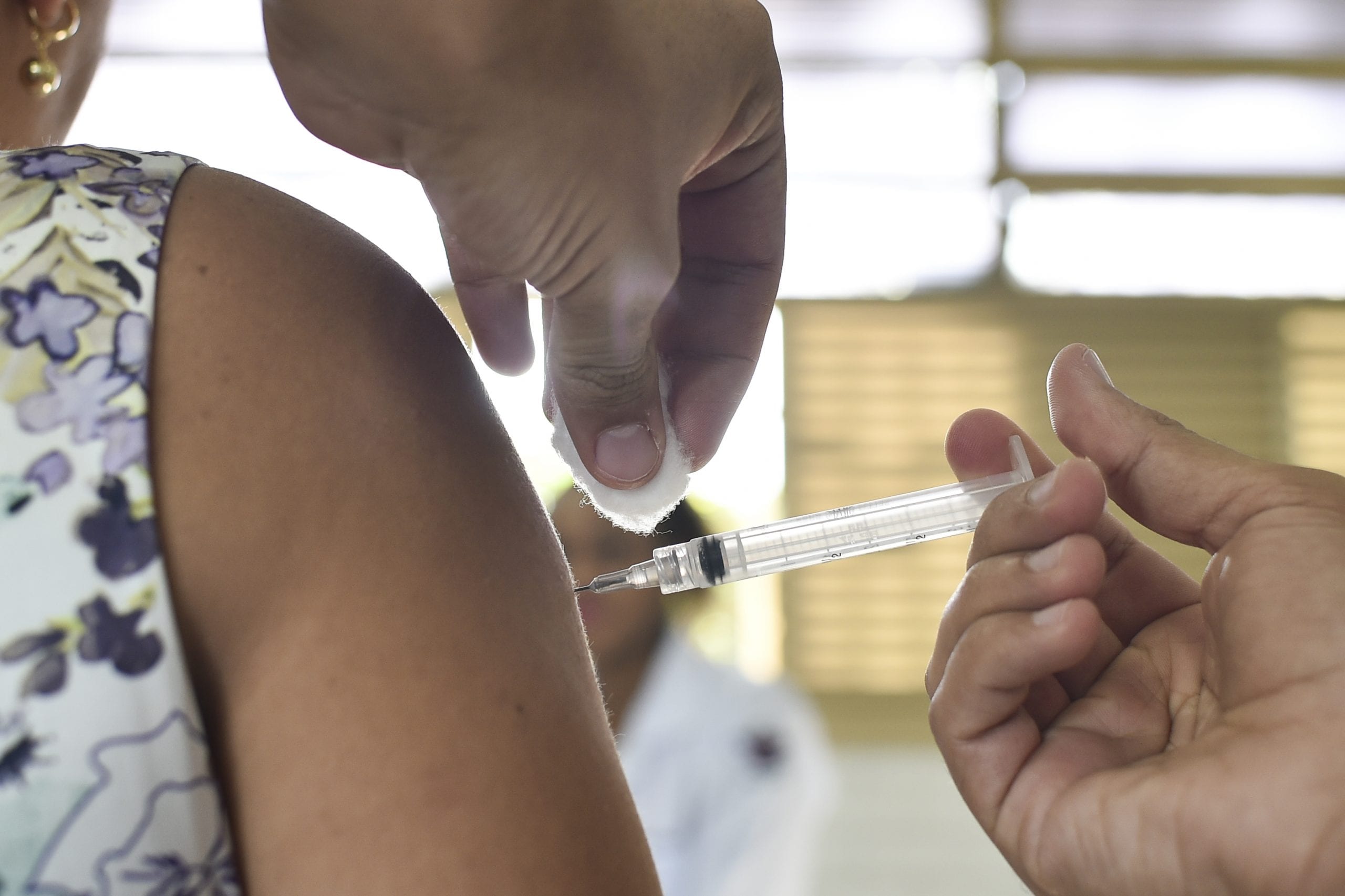
The most common way that flu shots are made is using an egg-based manufacturing process that has been around for > 70 years. In this instance, the Centers for Disease Control and Prevention (CDC) provide a manufacturer with flu viruses, which the manufacturer then injects into fertilized chicken eggs. The eggs are then incubated for several days to allow the viruses to replicate. Then, the virus-containing fluid is collected from the eggs. For flu shots, the viruses for the vaccine are then killed. The manufacturing process continues with purification and testing, and then the resulting vaccine is tested by the FDA on a lot-by-lot basis before approval. Just like that lengthy explanation, the process itself takes at least six months to produce a vaccine in large quantities! Plus, you need a helluva lot of eggs.
Despite having used egg-based production methods for years, scientists are now looking to new methods for harvesting the flu viruses. In 2016, the FDA approved a sole vaccine manufacturer to use cell-based vaccine viruses. Cell-based flu vaccine production does not require chicken eggs because the vaccine viruses are grown in animal cell lines. Unlike eggs, cells can be frozen for later use to grow large volumes of cells replicating viruses for vaccine production.
Lastly, in 2013 the FDA approved recombinant technology, this production method does not use chicken eggs or animal cell lines at all in the production process. Manufacturers instead harvest proteins directly from the flu virus and combine them with portions of another virus that grows well in insect cells. Both of these new flu shot technologies result in a faster, more flexible start-up for the flu vaccine manufacturing process. And, alternatives to egg-based products provide a vital option for people with egg allergies.
Regardless of which of these manufacturing strategies is used, vaccine makers must anticipate months in advance which strains of flu will circulate each year. A new so-called universal vaccine approach is under investigation in a two-year clinical trial involving more than 2,000 patients by researchers in Oxford, England. Unlike other flu vaccines that make use of the proteins on the surface of the virus, this new vaccine strategy works to fight all strains of the virus by using proteins found in the core of the virus, which stay largely the same across different strains. Researchers hope the new vaccine will provide better protection when used alongside the regular seasonal flu shot.
Any way you slice it, current recommendations show that an annual flu vaccine is the best way to reduce your risk of getting sick with seasonal flu and spreading it to others. Trust me, it won't be the thing your friends are thankful for this Thanksgiving! Still have questions? Go here to check out the approved influenza vaccines for the 2017-2018 flu season.
 Lydia Anderson is a Dual DVM-PhD graduate student at the University of Georgia. Since completing her PhD in Infectious Diseases, she has been working on her DVM at the College of Veterinary Medicine with an emphasis in public health and translational medicine. She plans to use her training to help address the questions and challenges facing One Health due to emerging and zoonotic infectious diseases. When she is not busy learning how to save all things furry and playing with test tubes, Lydia can be found either freestyle cooking for her friends and family or binge watching Netflix with her rescue pup, Luna. More from Lydia Anderson.
Lydia Anderson is a Dual DVM-PhD graduate student at the University of Georgia. Since completing her PhD in Infectious Diseases, she has been working on her DVM at the College of Veterinary Medicine with an emphasis in public health and translational medicine. She plans to use her training to help address the questions and challenges facing One Health due to emerging and zoonotic infectious diseases. When she is not busy learning how to save all things furry and playing with test tubes, Lydia can be found either freestyle cooking for her friends and family or binge watching Netflix with her rescue pup, Luna. More from Lydia Anderson.







